- Home
- /
- HTMA – Accurate Nutrient...
- /
- Taking an Accurate Hair...
How To Take A Sample
Proper hair sample collection is extremely important and cannot be overemphasised. Please read the instructions below.
We require 0.125 grams (125 mg)
1 teaspoon or enough to cover a 20-cent coin
Clean scalp hair, max 3cm long; cut excess hair.
Place hair in paper, NOT plastic!
SAMPLE COLLECTION PROTOCOL
Proper hair sample collection is extremely important and cannot be overemphasised. The following guidelines should be maintained to ensure the collection of a metabolically representative sample and to avoid the introduction of external contaminants prior to, during, and following the collection process.
Sample Type
Scalp hair is the only source recommended for analysis. Pubic and other body hair should only be used as a last resort if scalp hair is not available.
Note: Pubic and other body hair is only recommended for confirmation of elevated toxic metals found in the scalp hair and/or to rule out external contamination of the scalp hair.
Preparation
The portion of hair to be collected should be freshly washed with plain shampoo (definitely not medicated eg anti-dandruff), well rinsed and untreated, i.e. not permed, dyed or bleached.
If all of the hair has been chemically treated, wait until sufficient new virgin growth has emerged to allow collection. (Why not select small sections of hair at the back of the head that are hidden from view and skip doing the re-growth dye for a couple of months? )
The hair should also be free of all gels, oils, and hair creams prior to sample collection. For those individuals environmentally and/or occupationally exposed to external contaminants (welding, mining, etc.), special care should be taken to limit exposure between washing the hair and collecting the sampled hair.
Location
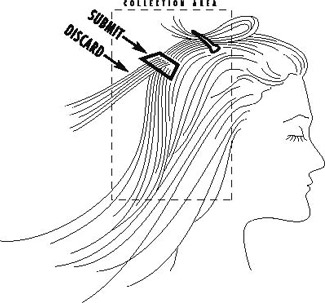
Each collected sample should be taken in small portions from at least four to five different locations on the scalp. The recommended areas for collection are the nape of the neck, posterior vertex and posterior temporal regions.
Note: All reference range correlations, dietary recommendations and interpretive report content assume that the complete hair specimen originated solely from the above-defined regions of the scalp.
Sample Length
High-grade stainless steel scissors or thinning shears should be used to cut the hair as close to the scalp as possible. The length of the collected hair should not exceed 3 – 4 cm. The proximal portion (3cm closest to the root) should be retained, and the excess discarded.
Note: The proximal portion is reflective of the most recent metabolic activity.
Weight
The weight requested for a hair specimen is 125 milligrams (0.125 Gram). Use of the Hair Weight Scale Cards will ensure the collection of sufficient sample weight. However, if a Hair Weight Scale is not available, one full teaspoon should approximate the weight requirement.
Packaging
Upon cutting the sample, the hair should be placed directly into a clean hair specimen envelope normally provided by the laboratory and then sealed with the glue flap only. Do not use plastic bags in place of the standard paper envelopes to hold the hair specimen. In addition, do not use staples, paper clips, adhesive tape, aluminum foil, or other metal and paper material of any kind to seal, secure or wrap the hair envelope and/or the hair specimen contained within.
Submittal Form
Each sample specimen must be accompanied by a standard HTMA Laboratory Submittal Form, completed in its entirety and signed by the health professional.
Test results and report will be available approximately 10 business days from the date we receive your hair sample and payment.
